 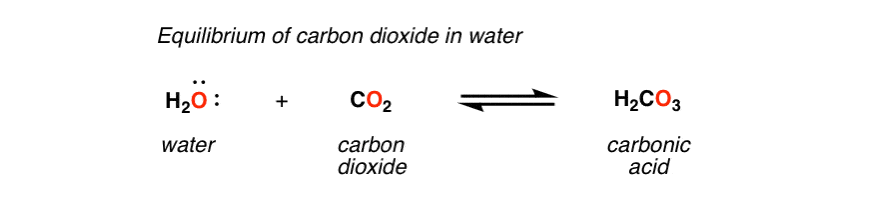
Ī high-pressure version of deuterated carbonic acid, i.e. Pure carbonic acid is mainly produced in two ways: the proton-irradiation of pure solid carbon dioxide and by reacting hydrogen chloride and potassium bicarbonate at 100 K in methanol. In the presence of water, it decomposes to form carbon dioxide and water, which further accelerates the decomposition. Carbon dioxide can be described as the anhydride of carbonic acid.ģ, is quite stable at ambient temperatures as a gas. 6.8, at equilibrium carbonic acid will be almost 50% dissociated in the extracellular fluid ( cytosol) which has a pH of ca. Strictly speaking the term "carbonic acid" refers to the chemical compound with the formula Hģ however, some biochemistry literature uses the term to incorrectly refer to dissolved carbon dioxide in extracellular fluid. Use of the term carbonic acid Speciation for a monoprotic acid, AH as a function of pH In physiology, carbon dioxide excreted by the lungs may be called volatile acid or respiratory acid. When Henry's law is used to calculate the value of the term in the denominator care is needed with regard to dimensionality. The value of log β 1 decreases with increasing ionic strength, I In the following expressions represents the concentration, at equilibrium, of the chemical species H +, etc. The stability constants database contains 136 entries with values for the overall protonation constants, β 1 and β 2, of the carbonate ion. It has been estimated that the increase in dissolved carbon dioxide has caused the ocean's average surface pH to decrease by about 0.1 from pre-industrial levels. Įxpected change refers to predicted effect of continued ocean acidification. The acidification of natural waters is caused by the increasing concentration of carbon dioxide in the atmosphere, which is caused by the burning of increasing amounts of coal and hydrocarbons. The Bjerrum plot shows typical equilibrium concentrations, in solution, in seawater, of carbon dioxide and the various species derived from it, as a function of pH. In aqueous solution carbonic acid behaves as a diprotic acid. 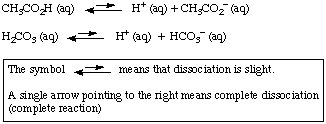
Chemical equilibria Equilibrium constant values Bjerrum plot for carbonate speciation in seawater (ionic strength 0.7 mol dm −3) In biochemistry and physiology, the name "carbonic acid" is sometimes incorrectly applied to aqueous solutions of carbon dioxide, which play an important role in the bicarbonate buffer system, used to maintain acid–base homeostasis. The interconversion of carbon dioxide and carbonic acid is related to the breathing cycle of animals and the acidity of natural waters. The molecule rapidly converts to water and carbon dioxide in the presence of water however, in the absence of water, contrary to popular belief, it is quite stable at room temperature. If the 25.00 mL sample that was titrated had been produced by dilution of a 10.In chemistry, carbonic acid is an organic compound with the chemical formula H 2 C O 3.If 37.63 mL of thiosulfate solution were needed to titrate a 25.00 mL sample of an acid, what was the H + ion concentration of the acid? The thiosulfate solution was determined to be 1.023 M. 
The endpoint is reached when the solution becomes colorless. The amount of iodine produced from this reaction can be determined by subsequent titration with thiosulfate: 2S 2O 3 2+ + I 2 → S 4O 6 2− + 2I − Starch is used as the indicator in this titration because starch reacts with iodine in the presence of iodide to form an intense blue complex. ♦ A method for determining the molarity of a strongly acidic solution has been developed based on the fact that a standard solution of potassium iodide and potassium iodate yields iodine when treated with acid: IO 3 − + 5I − + 6H + → 3I 2 + 3H 2O If the overall reaction sequence was 75% efficient, how many grams of carbonic acid were initially dissolved in the 250 mL to produce the calcium bicarbonate?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
